Emricasan
Emricasan (IDN-6556, PF-03491390) is a potent irreversible pan-caspase inhibitor with the ability to rapidly reduce elevated levels of serum ALT, AST and caspase mediated cleavage of cytokeratin-18 in HCV infected patients. 1,2,3 To date, emricasan has been studied in more than 550 individuals and has exhibited a safety profile similar to placebo.
Emricasan is currently in three Phase 2 clinical trials, including two trials in patients with liver cirrhosis. Here we report the effect of emricasan in healthy volunteers on measures of serum caspase activity and apoptosis. These results are compared with the effect of emricasan in subjects with varying degrees of hepatic impairment, as well as the effect of emricasan in subjects with severe renal impairment.
仅供研究使用。 我们不向患者出售。
化学信息
| 名称 | Emricasan |
|---|---|
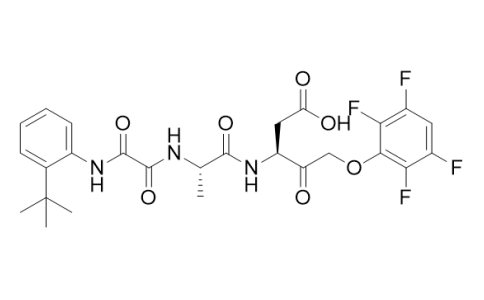
| Iupac 化学名称 | (S)-3-((S)-2-(2-((2-(tert-butyl)phenyl)amino)-2-oxoacetamido)propanamido)-4-oxo-5-(2,3,5,6-tetrafluorophenoxy)pentanoic acid |
| 同义词 | IDN-6556;IDN6556;PF 03491390;PF-03491390;Emricasan;PF03491390 |
| 英文同义词 | IDN-6556;IDN6556;PF 03491390;PF-03491390;Emricasan;PF03491390 |
| 分子式 | C26H27F4N3O7 |
| 分子量 | 569.51 |
| Smile | C(C)(C)(C)C1=C(C=CC=C1)NC(C(=O)N[C@H](C(=O)N[C@@H](CC(=O)O)C(COC1=C(C(=CC(=C1F)F)F)F)=O)C)=O |
| InChiKey | SCVHJVCATBPIHN-SJCJKPOMSA-N |
| InChi | InChI=1S/C26H27F4N3O7/c1-12(31-24(38)25(39)32-16-8-6-5-7-13(16)26(2,3)4)23(37)33-17(10-19(35)36)18(34)11-40-22-20(29)14(27)9-15(28)21(22)30/h5-9,12,17H,10-11H2,1-4H3,(H,31,38)(H,32,39)(H,33,37)(H,35,36)/t12-,17-/m0/s1 |
| Cas号 | 254750-02-2 |
| 相关CAS号 |
订购信息
| 包装 | 价格 | 库存 | 纯度 | 备货期 |
|---|---|---|---|---|
| 大货 | 询价 | 询价 | 询价 |
| 外观性状 | Off-white powder |
|---|---|
| 纯度 | 98% |
| 存储 | -20 ºC for 3 years |
| 可溶性 | Soluble in DMSO |
| 处理方式 | |
| 运输条件 | Shipped under ambient temperature |
| 海关编码 |
| Targets | |
|---|---|
| Mechanism | |
| Cell study | |
| Animal study | |
| Clinical study |
1. Frenette CT1, Morelli G2, Shiffman ML3, etc.Emricasan Improves Liver Function in PatientsWith Cirrhosis and High Model for End-Stage Liver Disease Scores Compared With Placebo. Clin Gastroenterol Hepatol. 2019 Mar;17(4):774-783.e4. doi: 10.1016/j.cgh.2018.06.012. Epub 2018 Jun 18.
2. Khurana S1, Khara HS1. Is Emricasan (IDN-6556), an oral pan-caspase inhibitor, ready for controlled trials? Hepatology. 2019 May 2. doi: 10.1002/hep.30683.