CPI-613(Devimistat )
编号: 51905
Cas号: 95809-78-2
纯度: ≧98.0%
CPI-613( Devimistat ) is an E1 pyruvate dehydrogenase (PDH) modulator that prevents cancer cells from metabolizing glucose for energy.
CPI-613( Devimistat ), Bulk in stock, contact us by email for Bulk quotation.
仅供研究使用。 我们不向患者出售。
化学信息
| 名称 | CPI-613(Devimistat ) |
|---|---|
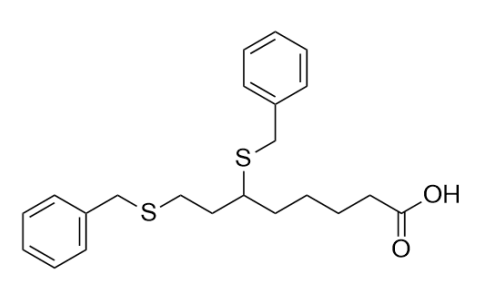
| Iupac 化学名称 | 6,8-bis(benzylthio)octanoic acid, OCTANOIC ACID, 6,8-BIS((PHENYLMETHYL)THIO)-, (+/-)-6,8-BIS(BENZYLTHIO)OCTANOIC ACID |
| 同义词 | CPI-613 ; CPI613 ; CPI 613 ; Devimistat |
| 英文同义词 | CPI-613 ; CPI613 ; CPI 613 ; Devimistat |
| 分子式 | C22H28O2S2 |
| 分子量 | 388.59 |
| Smile | O=C(O)CCCCC(SCC1=CC=CC=C1)CCSCC2=CC=CC=C2 |
| InChiKey | ZYRLHJIMTROTBO-UHFFFAOYSA-N |
| InChi | InChI=1S/C22H28O2S2/c23-22(24)14-8-7-13-21(26-18-20-11-5-2-6-12-20)15-16-25-17-19-9-3-1-4-10-19/h1-6,9-12,21H,7-8,13-18H2,(H,23,24) |
| Cas号 | 95809-78-2 |
| 相关CAS号 | 95809-78-2 |
订购信息
| 包装 | 价格 | 库存 | 纯度 | 备货期 |
|---|---|---|---|---|
| 大货 | 询价 | 询价 | 询价 |