Pimodivir ( VX-787 )
Pimodivir, also known as VX-787, JNJ-872, is a novel inhibitor of influenza virus replication that blocks the PB2 cap-snatching activity of the influenza viral polymerase complex. VX-787 binds the cap-binding domain of the PB2 subunit with a KD (dissociation constant) of 24 nM as determined by isothermal titration calorimetry (ITC). The cell-based EC50 (the concentration of compound that ensures 50% cell viability of an uninfected control) for VX-787 is 1.6 nM in a cytopathic effect (CPE) assay, with a similar EC50 in a viral RNA replication assay. VX-787 is active against a diverse panel of influenza A virus strains, including H1N1pdm09 and H5N1 strains, as well as strains with reduced susceptibility to neuraminidase inhibitors (NAIs).
For research use only. We do not sell to patients.
Chemical Information
| Name | Pimodivir ( VX-787 ) |
|---|---|
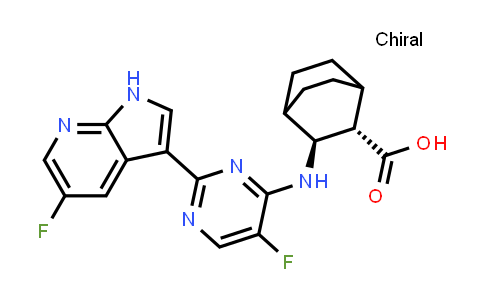
| Iupac Chemical Name | (2S,3S)-3-((5-Fluoro-2-(5-fluoro-1H-pyrrolo[2,3-b]pyridin-3-yl)pyrimidin-4-yl)amino)bicyclo[2.2.2]octane-2-carboxylic Acid |
| Synonyms | VX-787; VX 787; VX787; JNJ-63623872; JNJ63623872; JNJ 63623872; JNJ-872; JNJ 872; JNJ872; VRT-0928787; VRT 0928787; VRT0928787; pimodivir |
| Molecular Formula | C20H19F2N5O2 |
| Molecular Weight | 399.4018 |
| Smile | O=C([C@H]1C(CC2)CCC2[C@@H]1NC3=NC(C4=CNC5=NC=C(F)C=C54)=NC=C3F)O |
| InChiKey | JGPXDNKSIXAZEQ-SBBZOCNPSA-N |
| InChi | InChI=1S/C20H19F2N5O2/c21-11-5-12-13(7-24-17(12)23-6-11)18-25-8-14(22)19(27-18)26-16-10-3-1-9(2-4-10)15(16)20(28)29/h5-10,15-16H,1-4H2,(H,23,24)(H,28,29)(H,25,26,27)/t9?,10?,15-,16-/m0/s1 |
| CAS Number | 1629869-44-8 |
| Related CAS |
Ordering Information
| Packaging | Price | Availability | Purity | Shipping Time |
|---|---|---|---|---|
| Bulk | Enquiry | Enquiry | Enquiry |
| Formulation | Solid powder |
|---|---|
| Purity | 98% Min. |
| Storage | Dry, dark and at 0 - 4 C for short term (days to weeks) or -20 C for long term (months to years). |
| Solubility | Soluble in DMSO |
| Handling | |
| Shipping Condition | Shipped under ambient temperature as non-hazardous chemical. This product is stable enough for a few weeks during ordinary shipping and time spent in Customs. |
| HS Code |
| Targets | |
|---|---|
| Mechanism | |
| Cell study | |
| Animal study | |
| Clinical study |
1: Boyd MJ, Bandarage UK, Bennett H, Byrn RR, Davies I, Gu W, Jacobs M, Ledeboer MW, Ledford B, Leeman JR, Perola E, Wang T, Bennani Y, Clark MP, Charifson PS. Isosteric replacements of the carboxylic acid of drug candidate VX-787: Effect of charge on antiviral potency and kinase activity of azaindole-based influenza PB2 inhibitors. Bioorg Med Chem Lett. 2015 May 1;25(9):1990-4. doi: 10.1016/j.bmcl.2015.03.013. Erratum in: Bioorg Med Chem Lett. 2016 Jan 1;26(1):240. PubMed PMID: 25827523.
2: Byrn RA, Jones SM, Bennett HB, Bral C, Clark MP, Jacobs MD, Kwong AD, Ledeboer MW, Leeman JR, McNeil CF, Murcko MA, Nezami A, Perola E, Rijnbrand R, Saxena K, Tsai AW, Zhou Y, Charifson PS. Preclinical activity of VX-787, a first-in-class, orally bioavailable inhibitor of the influenza virus polymerase PB2 subunit. Antimicrob Agents Chemother. 2015 Mar;59(3):1569-82. doi: 10.1128/AAC.04623-14. PubMed PMID: 25547360; PubMed Central PMCID: PMC4325764.
3: Clark MP, Ledeboer MW, Davies I, Byrn RA, Jones SM, Perola E, Tsai A, Jacobs M, Nti-Addae K, Bandarage UK, Boyd MJ, Bethiel RS, Court JJ, Deng H, Duffy JP, Dorsch WA, Farmer LJ, Gao H, Gu W, Jackson K, Jacobs DH, Kennedy JM, Ledford B, Liang J, Maltais F, Murcko M, Wang T, Wannamaker MW, Bennett HB, Leeman JR, McNeil C, Taylor WP, Memmott C, Jiang M, Rijnbrand R, Bral C, Germann U, Nezami A, Zhang Y, Salituro FG, Bennani YL, Charifson PS. Discovery of a novel, first-in-class, orally bioavailable azaindole inhibitor (VX-787) of influenza PB2. J Med Chem. 2014 Aug 14;57(15):6668-78. doi: 10.1021/jm5007275. PubMed PMID: 25019388.
4: Smee DF, Barnard DL, Jones SM. Activities of JNJ63623872 and oseltamivir against influenza A H1N1pdm and H3N2 virus infections in mice. Antiviral Res. 2016 Dec;136:45-50. doi: 10.1016/j.antiviral.2016.10.009. PubMed PMID: 27771390.
5: Stevaert A, Naesens L. The Influenza Virus Polymerase Complex: An Update on Its Structure, Functions, and Significance for Antiviral Drug Design. Med Res Rev. 2016 Nov;36(6):1127-1173. doi: 10.1002/med.21401. Review. PubMed PMID: 27569399; PubMed Central PMCID: PMC5108440.
6: Fu Y, Gaelings L, Söderholm S, Belanov S, Nandania J, Nyman TA, Matikainen S, Anders S, Velagapudi V, Kainov DE. JNJ872 inhibits influenza A virus replication without altering cellular antiviral responses. Antiviral Res. 2016 Sep;133:23-31. doi: 10.1016/j.antiviral.2016.07.008. PubMed PMID: 27451344.
Chemical Structure