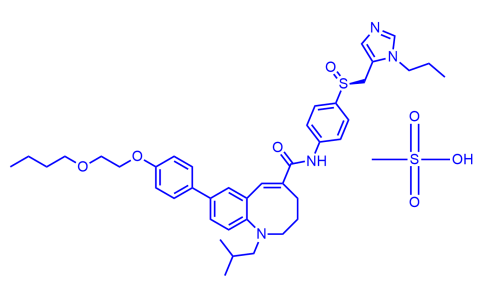
Cenicriviroc Mesylate
Cenicriviroc (CVC) is an oral, once-daily, potent immunomodulator that blocks two chemokine receptors, CCR2 and CCR5, which are intricately involved in the inflammatory and fibrogenic pathways in NASH known to cause liver damage and often lead to cirrhosis, liver cancer, or liver failure. Because of this unique mechanism of action, targeting two of the main engines driving NASH, Cenicriviroc (CVC) has the potential to play a differentiated role in the management of adults with liver fibrosis due to NASH and may form the cornerstone of NASH combination treatment strategies, both as a single agent and in combination with other agents targeting metabolic pathways. Cenicriviroc (CVC) has been granted Fast Track status in adults with liver fibrosis due to NASH, the patient population at highest risk of progression to cirrhosis.
Cenicriviroc(Cas No.: 497223-25-3) is in stock.
For research use only. We do not sell to patients.
Chemical Information
| Name | Cenicriviroc Mesylate |
|---|---|
| Iupac Chemical Name | (S,E)-8-(4-(2-Butoxyethoxy)phenyl)-1-isobutyl-N-(4-(((1-propyl-1H-imidazol-5-yl)methyl)sulfinyl)phenyl)-1,2,3,4-tetrahydrobenzo[b]azocine-5-carboxamide, mesylate |
| Synonyms | Cenicriviroc Mesylate,TAK-652 Mesylate; TBR-652 Mesylate,TAK652,TAK 652 |
| Molecular Formula | C42H56N4O7S2 |
| Molecular Weight | 793.05 |
| Smile | O=C(/C1=C/C2=CC(C3=CC=C(OCCOCCCC)C=C3)=CC=C2N(CC(C)C)CCC1)NC4=CC=C([S@](CC5=CN=CN5CCC)=O)C=C4.OS(=O)(C)=O |
| InChiKey | IXPBPUPDRDCRSY-YLZLUMLXSA-N |
| InChi | InChI=1S/C41H52N4O4S.CH4O3S/c1-5-7-22-48-23-24-49-38-15-10-32(11-16-38)33-12-19-40-35(25-33)26-34(9-8-21-44(40)28-31(3)4)41(46)43-36-13-17-39(18-14-36)50(47)29-37-27-42-30-45(37)20-6-2;1-5(2,3)4/h10-19,25-27,30-31H,5-9,20-24,28-29H2,1-4H3,(H,43,46);1H3,(H,2,3,4)/b34-26+;/t50-;/m0./s1 |
| CAS Number | 497223-28-6 |
| Related CAS |
Ordering Information
| Packaging | Price | Availability | Purity | Shipping Time |
|---|---|---|---|---|
| Bulk | Enquiry | Enquiry | Enquiry |
| Formulation | pale yellow solid |
|---|---|
| Purity | 98% Min. |
| Storage | Dry, dark and at 0 - 4 C for short term (days to weeks) or -20 C for long term (months to years). |
| Solubility | Soluble in DMSO, not in water |
| Handling | |
| Shipping Condition | Shipped under ambient temperature |
| HS Code |
| Targets | |
|---|---|
| Mechanism | |
| Cell study | |
| Animal study | |
| Clinical study |
1. Neokosmidis G, Tziomalos K., Role of cenicriviroc in the management of nonalcoholic fatty liver disease.World J Gastroenterol. 2018 Dec 28;24(48):5415-5417. doi: 10.3748/wjg.v24.i48.5415
2. Covino DA, Purificato C, Catapano L, Galluzzo CM, Gauzzi MC, Vella S, Lefebvre E, Seyedkazemi S, Andreotti M, Fantuzzi L., APOBEC3G/3A Expression in Human Immunodeficiency Virus Type 1-Infected Individuals Following Initiation of Antiretroviral Therapy Containing Cenicriviroc.Front Immunol. 2018 Aug 8;9:1839. doi: 10.3389/fimmu.2018.01839. eCollection 2018.
3. D'Antoni ML, Mitchell BI, McCurdy S, Byron MM, Ogata-Arakaki D, Chow D, Mehta NN, Boisvert WA, Lefebvre E, Shikuma CM, Ndhlovu LC, Baumer Y., Cenicriviroc inhibits trans-endothelial passage of monocytes and is associated with impaired E-selectin expression. J Leukoc Biol. 2018 Dec;104(6):1241-1252. doi: 10.1002/JLB.5A0817-328RRR. Epub 2018 Aug 8.
Chemical Structure